- Registration
- Welcome and Workshop Introduction by Charles River Laboratories
- Contaminants, Causes, Investigation and Case Studies
Ziva Abraham - Refreshments
- Risk Mitigation with Modern Technologies (RMM, NGS
Christian Scheuermann - Difficult to Resolve LAL Investigations
- Lunch
- Putting it All in Practice: Group Breakouts
- Knowledge Sharing (Kahoot game)
Topic Details
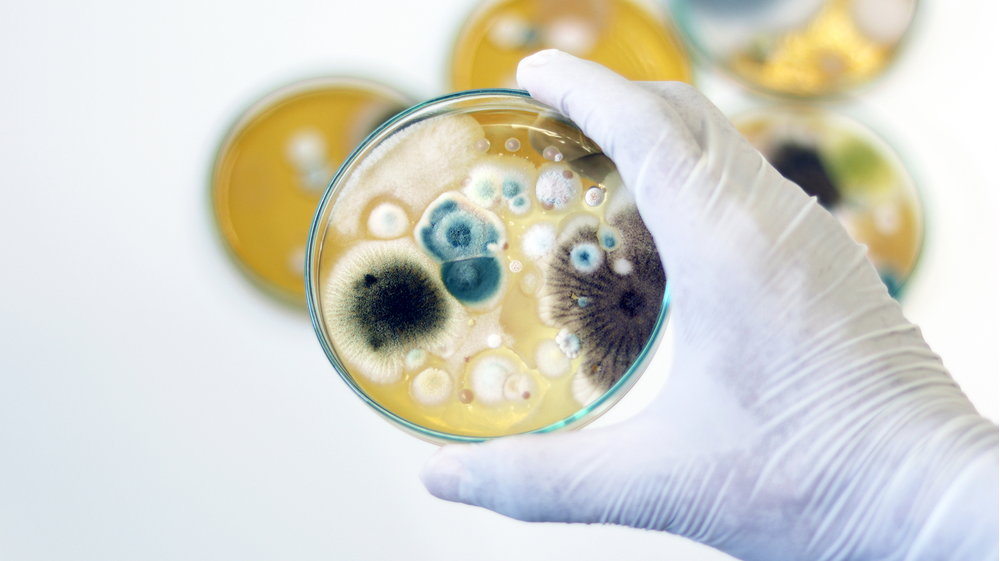
Mold Investigations
Mold remediation is a daunting task. More often, than not, remediation efforts fail. Mold can be removed temporarily but often returns shortly thereafter. These investigations must be thorough, with a solid understanding of the species involved, their associated sporulation patterns, and their susceptibility to disinfection or sterilization.
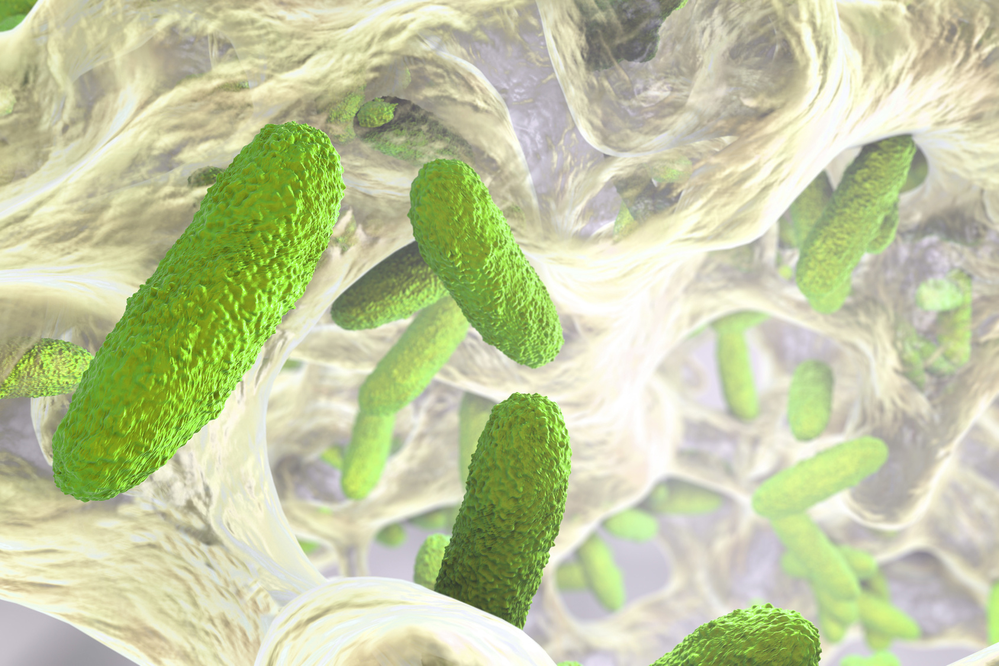
Biofilm Investigations
Often biofilm issues in the water systems or process equipment are blamed on sampling or aseptic techniques. Excursions in any one of the critical parameters (conductivity, TOC, bioburden, and endotoxin) can sometimes be transient by nature. In order to conduct a thorough investigation, you must understand the various types, the forming organisms, the recovery process by testing methods used, and its structure. Additionally, biofilm can be tested against biocidal agents or disinfectants in the laboratory setting instead of trial-and-error methods only to discover the biofilm persists. We will discuss how to understand result patterns derived by various parameters, how a comprehensive ID library aids investigations, and when to employ strain-typing for better result resolution.

Viable but Non-Culturable Organism Proliferation
Are microorganisms that are viable but non-culturable truly incapable of proliferation? Is so, you need to evaluate if they are posing any potential risks to pharmaceutical products or the pharmaceutical water system. We'll walk you through how and why biofilms continually build up in heated water systems and what are the patient-safety or contamination control risks posed by these microorganisms. Dive deeper into the meaning of this term, understand why this happens and how these microorganisms typically present. Learn how modern technologies such as rapid microbiological methods (RMM) and Next Generation Sequencing (NGS) can play an important role in mitigating these missed risks.

